risk stratification of patients
with Barrett's esophagus

A methylation-based approach to risk stratification
Esopredict® is a risk-stratification test offered in a small number of situations where the TissueCypher spatialomics approach is unable to provide a result. Esopredict uses a methylation-based assay that does not utilize fluorescence or tissue architecture, to determine a patient’s risk of progressing from non-dysplastic, indefinite or low grade Barrett’s esophagus (BE) to high-grade dysplasia (HGD) or esophageal adenocarcinoma (EAC). Esopredict is validated for patients with Barrett’s esophagus (BE) diagnosed as non-dysplastic, indefinite for dysplasia, or low-grade dysplasia.
How it Works
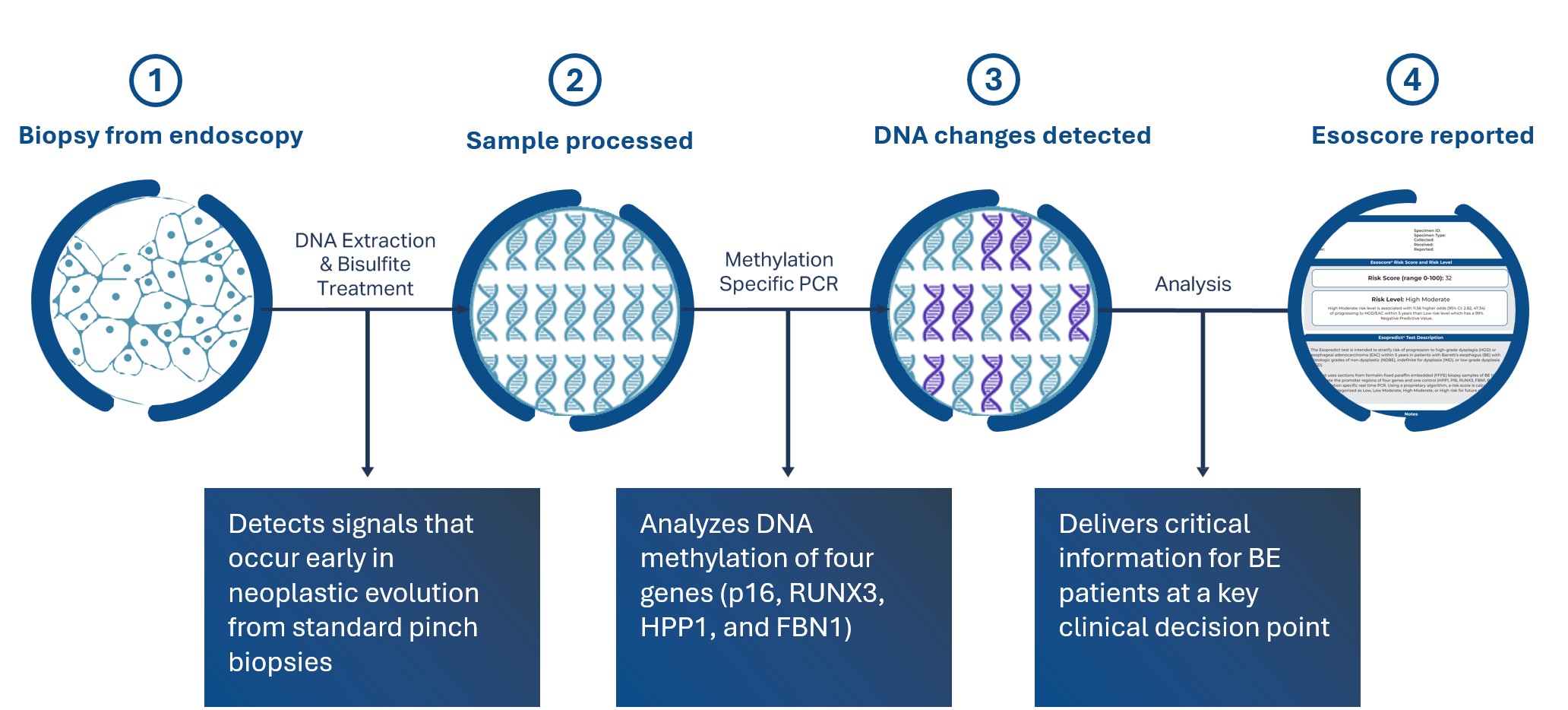
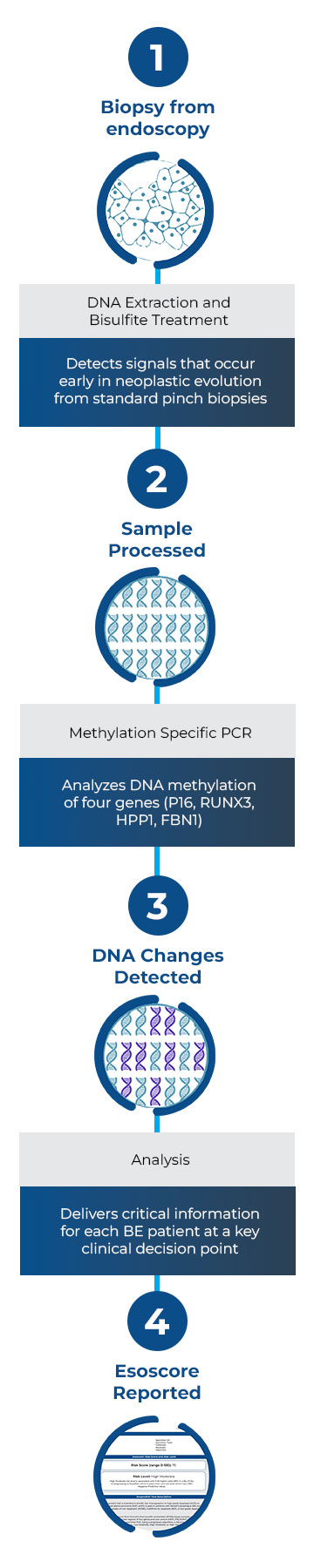
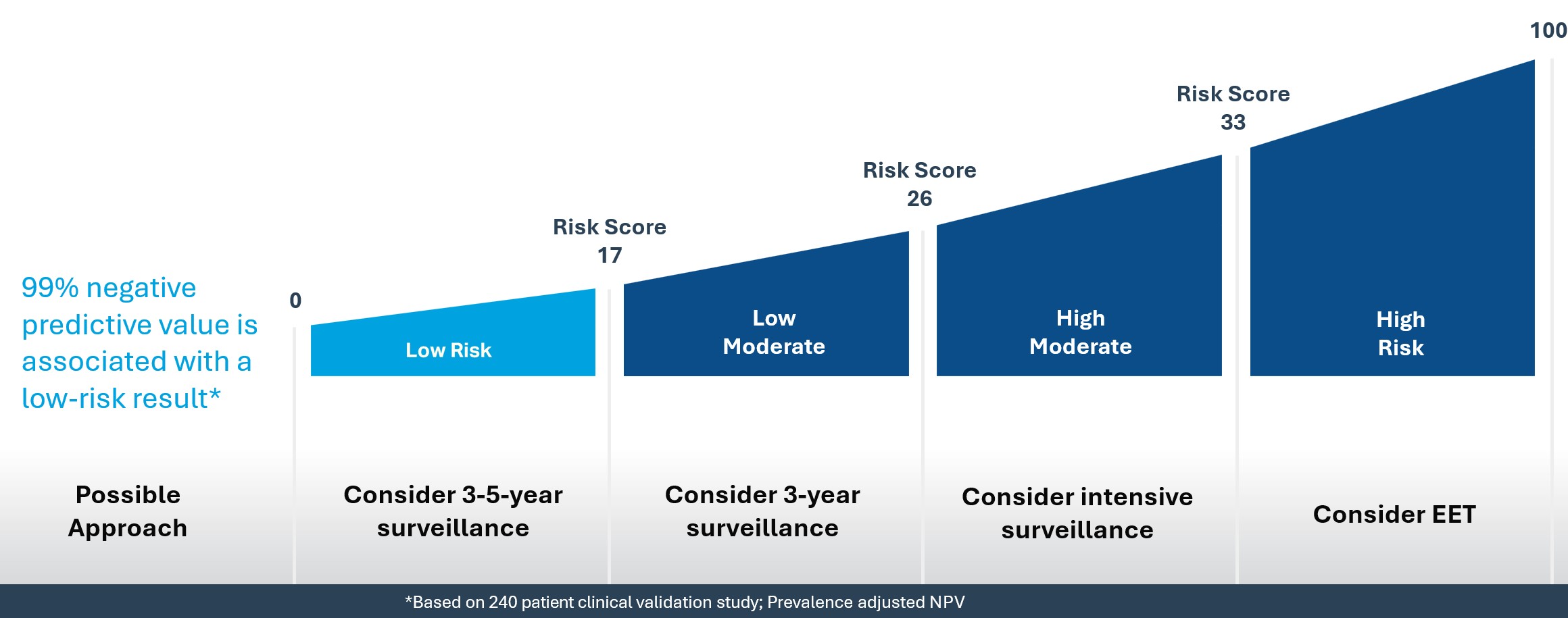
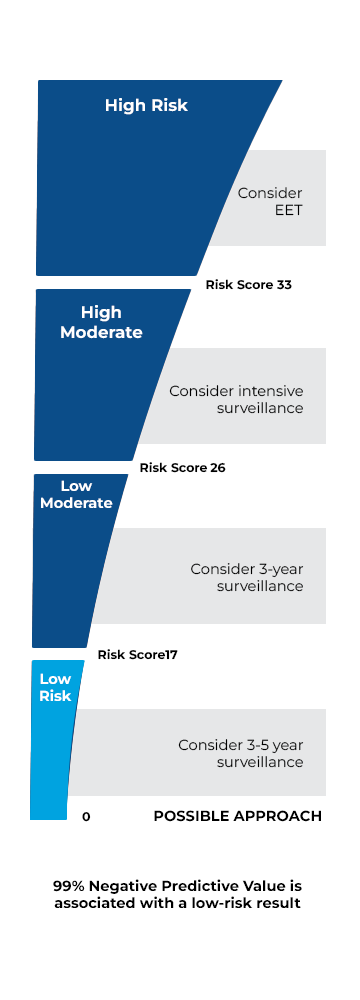
Esopredict quantifies the DNA methylation status of four genes: p16, RUNX3, HPP1, and FBN1. A risk prediction algorithm combines these methylation markers with the patient’s age to calculate their risk of progressing to HGD or EAC within 5 years. The results include a risk score from 0-100 and a risk level of low, low-moderate, high-moderate, or high.
Scientific Evidence
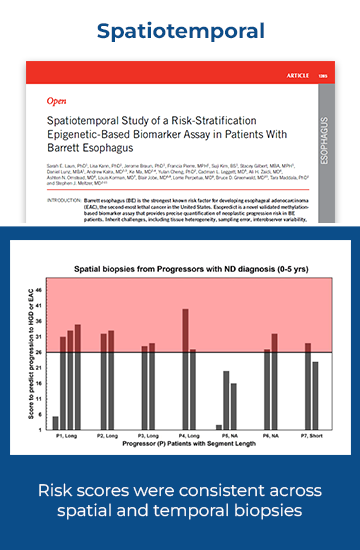
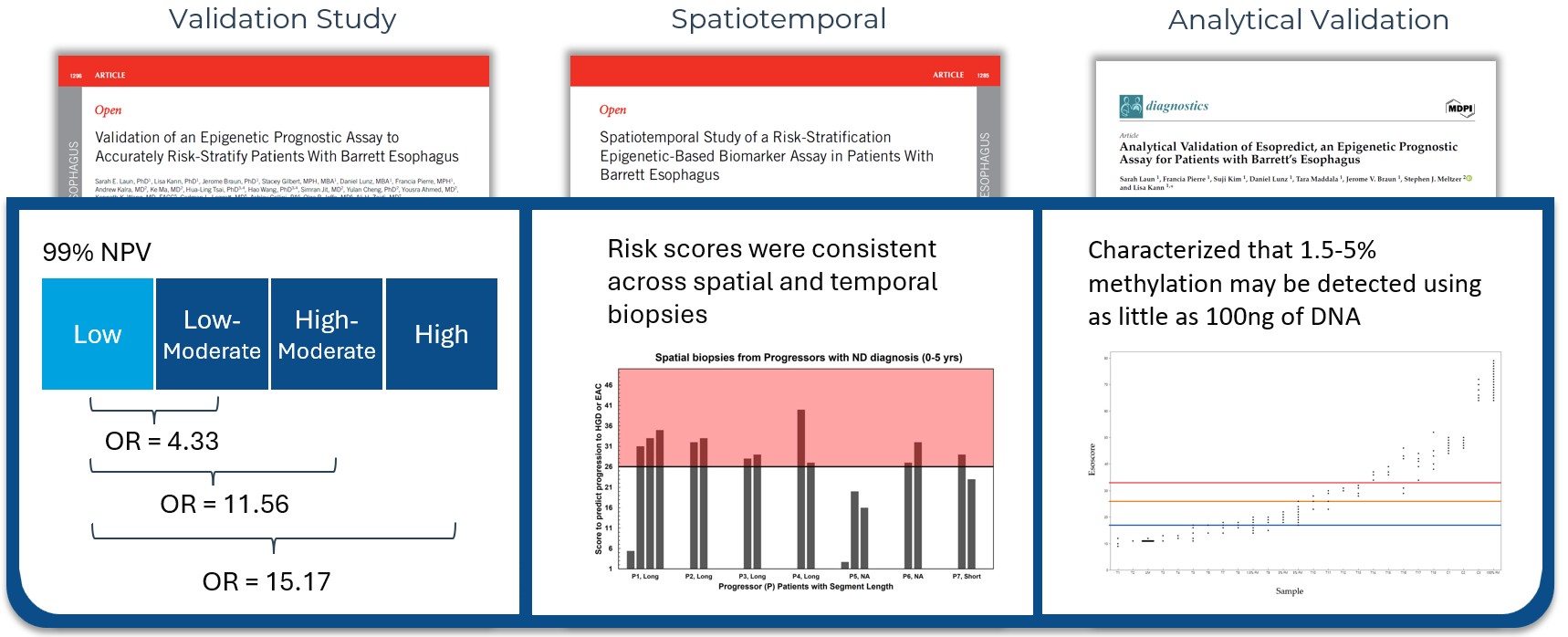
Esopredict test performance has been validated in three peer-reviewed publications. The data analyzed in those publications demonstrated a 99% negative predictive value (Laun SE, et al. Am J Gastroenterol. 2024.) and consistency across spatial and temporal biopsies (Laun SE, et al. Am J Gastroenterol. 2025.). Furthermore, the test was validated to show that 1.5 – 5% methylation may be detected using as little as 100ng of DNA (Laun SE, et al. Diagnostics (Basel). 2024.).
When is Esopredict offered?
TissueCypher is Castle’s primary risk stratification test for Barrett’s esophagus. In the small number of situations where the TissueCypher spatialomics approach is unable to provide a result, a “not reportable” result may be issued. In these cases, Castle’s Clinical Services team will reach out to the treating clinician to determine if Esopredict testing is appropriate for this patient. If authorized, testing will begin with pinch biopsy tissue already on hand, or if needed, Castle will reach out directly to the pathology laboratory to obtain additional tissue. After sufficient biopsy tissue is received, Esopredict results will be issued in approximately 10 days.

Pre-filled order
If TissueCypher is unable to provide a test result, then Castle will reach out via secure online portal or fax to alert the treating clinician of the option to have Esopredict testing performed

Clinician signature
Approve the Order Authorization in the portal or send a signed version via secure email or fax: (878) 213-3022
Castle obtains sample
If the tissue sample is at Castle, additional slides will be sectioned (8 x 5μm slides). If necessary, Castle Clinical Services will coordinate with your pathology lab to obtain additional tissue.

Esopredict results
The tissue sample is analyzed, and the patient's Esopredict results are delivered via the secure portal and/or your fax or secure email preferences.
Interpreting the Esopredict results
The Esopredict report provides both a Risk Level and a Risk Score to help guide patient management.

Risk Level
The risk level can be one of four categories: low, low-moderate, high-moderate, or high.

Risk Score
The risk score places the patient's risk on a 0-100 scale, with 100 representing the greatest risk.
Using the Esopredict risk level, a clinician may consider adjusting care that best fits with the patient’s individual risk. The Esopredict clinical validation study showed patients receiving low-risk results were at extremely low risk of progression with a 99% negative predictive value (Laun SE, et al. Am J Gastroenterol. 2024.). In comparison, a patient receiving a high-risk result was shown to be at 15x higher risk than a patient scoring low-risk, and such elevated risk may warrant consideration of shorter interval surveillance or endoscopic eradication therapy.
Frequently Asked Questions

A head-to-head comparison of the performance of TissueCypher to Esopredict has not been completed. TissueCypher has been studied more extensively, with 17 published studies, pooled analyses, and a systematic reviews and meta-analysis that evaluated 699 patients with known outcomes. Esopredict has been less well studied, having three published studies that analyzed 240 patients with known outcomes. Esopredict’s validation study showed a 99% negative predictive value for patients scoring low risk, while patients scoring high-risk were 15 times more likely to progress patients scoring low-risk. Castle is actively researching both tests as well as the potential combination of these tests to further improve risk stratification.

Esopredict runs on the same pinch biopsies as TissueCypher. Castle may have enough tissue on hand to perform Esopredict without reaching to the pathology laboratory. This is dependent upon how many slides were received to perform TissueCypher or if the block was initially sent. In cases where there is insufficient tissue to perform Esopredict, then Castle’s clinical services team will reach out to the pathology laboratory to obtain additional tissue for testing.

Yes, the treating clinician will need to sign the Esopredict requisition form. However, Castle will prefill the Esopredict requisition with the information received on the TissueCypher requisition form. Thus, the only item that may be needed is an electronic or physical signature authorizing the test.

Esopredict testing is only performed on biopsies from one level in both short- and long-segment patients.

No, the insurance company is only billed after a successful result is reported to the clinician. A not reportable test report does not result in a bill to insurance or an EOB.
To learn more about Esopredict,
please contact us